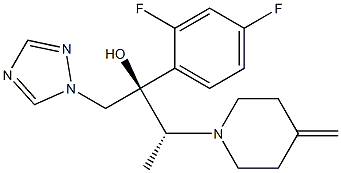
Efinaconazole is a triazole antifungal active pharmaceutical ingredient supplied as a white to off-white or beige crystalline powder for use in pharmaceutical formulations. It exerts its antifungal effect by selectively inhibiting fungal CYP51 (lanosterol 14α-demethylase), a key enzyme in ergosterol biosynthesis, disrupting fungal cell membrane integrity and inhibiting the growth of dermatophytes responsible for onychomycosis (fungal nail infections).
Efinaconazole is distinguished from earlier azole antifungals by its exceptionally low keratin binding affinity, which enables superior penetration through the nail plate and subungual space, a pharmacokinetic advantage that makes it particularly well suited for topical nail solution formulations targeting dermatophyte-caused onychomycosis. The API is packaged in double LDPE bags within HDPE drums or equivalent pharma-grade, moisture-protective containers to maintain stability under recommended storage conditions.
As a leading distribution partner, Molkem Chemicals ensures reliable supply of products aligned with industry quality standards and regulatory expectations. Each product is delivered as per agreed specifications, supported by standard documentation including COA, SDS, and TDS. If required, our technical team can provide additional stability data and regulatory assistance.